THE GREAT SALK VACCINE FIASCO Misuse of statistics, blackout of vaccine cases, cited by eminent Chicago doctor
https://www.vaclib.org/basic/polio/salk-fiasco.htm
THE GREAT SALK VACCINE FIASCO
Misuse of statistics, blackout of vaccine cases, cited by eminent Chicago doctor
THE GREAT SALK VACCINE FIASCO
Misuse of statistics, blackout of vaccine cases, cited by eminent Chicago doctor
By ERNEST B. ZEISLER, M.D.
(179 East Lake Shore Drive,
Chicago 11, Ill.)
Copyright, 1960, The Minority of One, Inc.
The Salk poliomyelitis vaccine was developed in about 1953 by Jonas Salk, who maintained that his vaccine contained only inactive virus and was effective and absolutely safe. He was able to convince those who urged caution that the "expensive and difficult procedures which had been suggested for the detection of possible residual live virus"(2) in his vaccine should be dispensed with; he based his assurance that this was entirely safe on a theoretical argument which has been severely criticized.
On April 12, 1955, results of a 1954 field test were published and the Salk vaccine became a licensed product. Prof. Paul Meier of the School of Hygiene and Public Health at Johns Hopkins University revealed that "the vaccines used in the field trial, which were produced by two of the manufacturers, had been extensively tested in three laboratories and had been found negative for live virus. Many of the lots of vaccine released after the field trial had been produced by other manufacturers and had been tested only by the producer. Therefore, the safety of these lots could not properly be judged from the results of the field trial.
All manufacturers had rejected some lots because live virus had been found in them, and therefore Salk's theory that safety was guaranteed by the method of preparation obviously did not apply." (3) The field trial itself had violated the cardinal principles of scientific procedure. As said by Brownlee in the Journal of the American Statistical Association: ". . . 59 per cent of the trial was worthless because of the lack of adequate controls. The remaining 41 per cent may be all right but contains internal evidence of bias in favor of the vaccinated. .. The reviewer . . . would point out that gamma globulin was triumphantly proclaimed effective by the National Foundation after a similar trial . . ." (4) (It may be of interest to note that in May of 1954, several months after it had been shown to be valueless in preventing poliomyelitis, the U.S. Public Health Service continued to recommend and distribute gamma globulin "for use against poliomyelitis"(5) No review of Brownlee's critique ever appeared in the Journal of the American Medical Association. The official report of the field trial was, nevertheless, used "by the National Foundation for Infantile Paralysis to push the vaccine into mass use in the spring of 1955." (6)
And so the public was deceived into permitting mass vaccination of children with a vaccine which should have been known to be unsafe and which was not known to be of any value in preventing poliomyelitis. Within two weeks it could no longer be denied that certain lots of vaccine had produced a number of cases of polio myelitis, and within another four weeks all the vaccine was withdrawn from use.
On November 15, 1955, the U.S. Public Health Service "released a report declaring that a single inoculation of the Salk vaccine used in 1955 was sufficient to give from 50 to 80 per cent protection against paralytic poliomyelitis; two days later it issued another report stressing the safety of the current Salk vaccine.
Dr.. Herbert Ratner, Associate Clinical Professor of Preventive Medicine and Public Health at the Stritch School of Medicine of Loyola University, and Health Commissioner of Oak Park, Ill., commented as follows: "The widespread national publicity that followed these reports naturally led the public and the medical profession at large to believe we now had a safe and highly effective vaccine.
"However, what was not made sufficiently clear in the reports and press stories that covered the country was thatthe first report, stressing excellent effectiveness, referred to an earlier model of a Salk vaccine and Hurt the second report, stressing current safety referred to a later model, . . . the Salk vaccine, for which great effectiveness is claimed on the basis of one inoculation, is a product that is no longer on the market nor in the hands of physicians . . . The Salk vaccine, then, which we were encouraged to believe is both highly effective and safe on the basis of recent reports, turns out to be, when highly effective, a vaccine that is no longer on the market and, when safe, a vaccine that has yet to make its appearance and clinically prove its effectiveness . . . during the summer the promoters of the vaccine continued to urge mass inoculations in spite of recognized ignorance on their part."(7)
In June of 1956, polio cases began to increase sharply in Chicago. Dr. Herman Bundesen, President of the Chicago Board of Health, was quoted as saying: "It's too early to speculate on the efficacy of the vaccine."(8) This moment of candor was not to recur from then until now. On the same day, Dr. John B. Hall, director of the Cook County Board of Health, said, concerning six cases of polio in children who had received, the Salk vaccine. he did not think the vaccine caused the polio attacks in those who got the disease after inoculation."(9) On July 3, 1956 the National Founda-tion for Infantile Paralysis sent a letter to all physicians over the signature of Hart E.Van Riper, its Medical Director, urging them to "give reassurance that the present Salk vaccine is safe and effective to patients, parents and others in your community who still needlessly doubt it ... the vaccine is at least 75% effective in preventing paralytic poliomyelitis. . . . Won't you take leadership in your community and among your patients to see that they get this safe, highly effective vaccine now?"
On July 9, I wrote to Dr. Van Riper, quoting Drs. Bundesen and Hall, and asking: "Why, if the vaccine has been proved to be 75% effective is it still too early even to speculate about its effectiveness? And why, if it has been proved safe is it possible for the head of a health department merely to think that it did not cause infection?" In his reply, dated July 12, Dr. Van Riper said as to Dr. Bundesen's remark that "it's too early to speculate about the effectiveness of the vaccine": "I can only assume that Doctor Bundesen intended to imply that we could expect an even greater degree of effectiveness in the prevention of paralytic poliomyelitis in 1956 as compared with 1955, . . ." That this is not at all what Dr. Bundesen intended to imply is shown by the fact that only two days after expressing his doubt, he called a conference of health authorities to decide whether or not vaccination with the Salk vaccine should not be entirely discontinued in view of the accelerated rise of new cases of paralytic polio in Chicago. Dr. Bundesen obviously was considering the possibility that the Salk vaccine would help spread the disease.
On July 27, there were already 203 reported cases of paralytic polio in Chicago. But Dr. Bundesen said: ". . . there were no paralytic cases among children who had received all three shots."(10) It must be obvious that this statement, even if true, was entirely without significance unless there was anyone in the affected area who had received all three injections. Compare the last six lines from The Walrus and the Carpenter:
"O Oysters,"-said the Carpenter,
"You've had a pleasant run!
Shall we be trotting home again?"
But answer came there none—
And this was scarcely odd, because
They'd eaten every one.
On the same day I wrote Dr. Van Riper the following: "If no child in the area had received three injections, then the fact that none of those with paralytic polio had received three doses is irrelevant and inevitable. In the daily figures which have been given there is always the statement as to how many of those who have come down with paralytic polio had been vaccinated, but never any figure as to how many children in the area had been vaccinated compared to the total number in the area. . . . People are being urged to have their children vaccinated at once, and physicians are urged to further this, with the implication that such procedure will be effective in stemming the tide of the present epidemic. . . . Yet, inasmuch as the third dose is to be given seven months after the first, only the first two could have' any possible effect this year."
This letter elicited only double-talk from Dr. Van Riper in a letter dated August 9. Dr. Bundesen continued to issue reassuring statements. On August 9 he stated he was "concerned with the drop off in the number of persons returning for their second shots of vaccine . . . The situation may become critical unless parents bring their children in for their second and third shots when they are due, and for the first inoculation if they have not already had it." "Of the city's 371 paralytic cases— the form against which the vaccine is effective—not a single case has been reported for any person who had the recommended three inoculations. There have been 54 among those getting only one and 13 among those with two."(11) The obvious explanation tor this division of the incidence of paralytic polio was, of course, that there were more persons who had had one injection than two, and perhaps nonewho had had all three. This was never suggested by the health authorities or by the medical societies or journals. Even Dr. Thomas Francis, Jr., of the University of Michigan School of Public Health, told the university's medical alumni: "Of all the 113 polio cases in Michigan diagnosed as paralytic, not one case has been reported among those children who had previously received three shots of vaccine."(12)On September 29, the U.S. Public Health Service said "three Salk shots have proved 100 per cent effective against polio so far this year." (13)
Still no indication that anyone had received all three injections! On September 30, I again wrote to Dr. Van Riper: "I have inquired from one of the foremost authorities in Chicago as to how many children in the Chicago epidemic area this summer had previously received -three injections. He said no one knew the answer, but that the number was certainly very small. I then asked him whether to his knowledge any of them had received all three injections, and he replied he did not know. I would greatly appreciate your reply to this question. . ." On October 26, after inconsequential interim correspondence, he finally answered: "I am sorry that to date there has not been sufficient time elapsed since the Chicago epidemic to enable anyone to give a definite answer to the question you have raised. I do know that a study is being made and feel sure this will be made public when it is completed." But no time was needed afterthe epidemic to determine how many persons had had all three injections beforethe epidemic began. In any case, here was a clear admission that no one knew so the repeated assurances of the 100 per cent effectiveness of three doses of the Salk vaccine in preventing paralytic polio in this epidemic, admit of no possible explanation other than either deliberate falsehood with intent to deceive or unconscionable stupidity.
By late November the public had seemingly become so apathetic about Salk vaccination that the pharmaceutical houses and the health authorities enlisted the aid of President Eisenhower, and on November 27 induced him to express alarm that there were 17,000,000 doses of Salk vaccine unused on the shelf and that they could "prevent paralysis or even death." The Sun-Timesquoted Dr. Bundesen as saying: "If everyone 45 or under gets the complete series, there will not be a single case of paralytic polio in Chicago in 1957"(14) thereby asserting that the vaccine in three doses was 100 per cent effective. "
On January 3, 1957, U.S. Public Health Service reported that paralytic polio in the United States had dropped from 10,641 cases in 1955 to 6,708 cases in 1956. This was a decrease of 37 per cent. The New York Times said "Health officials said the use of the Salk vaccine had undoubtedly reduced the disease but there was no way of knowing to what extent "(15)Immediately thereafter it said that in the same period hepatitis had decreased from 31,340 to 19,270 cases, a decrease of 38-1/2 percent, which was more than the decrease in paralytic polio. Health authorities said they had no explanation for this decrease. Later the same month it was reported at a meeting of the New York Academy of Sciences that there were records of more than 150 cases of paralytic polio, including several deaths, among persons persons who had received all three injections of Salk vaccine. Without ever referring to this, newspapers, medical journals and medical societies continued to plug for the vaccine. Members of the Illinois State Medical Society were circularized by Dr. Harold M. Camp, its secretary, with a letter dated February 11, 1957: "It is up to the medical profession to see that the public is advised immediately concerning the urgency of having every person between the ages of six months and 40 years protected ... all physicians should impress upon their patients that now is the time to develop the immunity offered by the three doses of vaccine."
In January of 1957. Tommy Mohr. a child from Albuquerque, N.M., contracted polio four months after his third inoculation with Salk vaccine; it was confirmed at Johns Hopkins Hospital that the death was due to polio. After a delay of six weeks this was reported by the National Foundation for Infantile Paralysis, which said that he had been "the first child positively known to have died of polio after three Salk vaccine shots."(16) At about this time Dr. Herbert Ratner began to wage what was practically a single-handed crusade against the Salk vaccine fraud. On February 17, 1957, he delivered a letter to four major Chicago daily newspapers, in which he said among other things: "The people of Metropolitan Chicago and the good name of medical science deserve something better than the campaign of fear based on misleading data presently being con-ALTERNATIYE VACCINE IS RECOMMENDED
U.S. Public Health Service finally has approved use of a live virus polio vaccine developed by Dr. Albert Sabin of Cincinnati. This vaccine won AM A endorsement at the annual meeting in Miami in June. The resolution's sponsor, Dr. C. C. McLean of Birmingham, Ala., pointed out the vaccine has been administered to millions of persons in 19 countries without producing a single proven case of paralytic polio. In the Soviet Union a program this year aimed at vaccination of all Russians between the ages of two months and 20 years with the attenuated virus vaccine.
ducted by the Chicago Health Commissioner in his promotion of Salk vaccination against poliomyelitis. The following facts have not as yet been made clear to the public or to the medical profession at large:
" 1. The use of vaccine in Chicago last summer . . . was an unequivocal failure. . . The proof of the failure is found in a comparison of the ratio of paralytic cases (the type of polio against which the vaccine is allegedly effective) to total polio cases in vaccinated and non-vaccinated groups. Paralytic polio was no lower proportionately in vaccinees than in non-vaccinees. In both groups the incidence was about 61%. This is in sharp contrast with Bundesen's contention in 1955, when mostly one shot was given, that the vaccine was 90% effective in paralytic polio.
"2. Contrary to Bundesen's implication, the chief reason individuals with one shot had more polio in 1956 than individuals with two shots is because there were more individuals who had one shot. As more individuals were vaccinated last summer, more who were vaccinated came down with polio. By July 19 one of every 13 cases was in vaccinee. By August 1, one out of seven was in a vaccinee, and from August 2 on, one out of every two cases of polio was in a vaccinee. . ."
"5. Neither the public nor the medical profession has been told that the bulk of the Salk vaccine now on the market is of poor potency. . . New York state authorities . . . reported a 600-fold variation in the potency of commercial vaccines. . .
"6. No respectable epidemiologist can agree with Bundesen when he states that if we do not intensively vaccinate 'The alternative is another polio outbreak this summer.' The fact is that the incidence of polio naturally decreases following a high incidence year. 1953 iIlustrates this. It showed a drop of 42% in the incidene of polio compared to l952." The Chicago Tribune, Chicago American, Chi-cago Sun-Times published none of the contents of this letter, whereas the Chicago Daily News, after three weeks, published a brief and watered-down ver-sion(17)and in the same issue continued to urge inoculation.
Dr. A. D. Langmuir of the Communicable Disease Center of U.S. Public Health Service had said at a meeting in Atlantic City in November of 1956 that the use of the vaccine in Chicago in 1956 "was a great disappointment."
Dr. Ratner's view of the matter is in accord with that of the Expert Committee on Poliomyelitis of the World Health Organization, which in its technical report in 1958 said: "It was noted in the Union of South Africa and in the USA, especially in the course of severe outbreaks in Hawaii and Chicago, that vaccination in the face of an epidemic did not appear to shorten its course. Laboratory and field studies have shown that vaccination does not prevent infection or interfere with dissemination of virus in the community. . ."(18)
In February of 1957, the National Foundation for Infantile Paralysis issued a pamphlet for physicians,(19)purporting to give information concerning the Salk vaccine, but actually giving much misinformation. They reported what they called a study(20)of 3,198 acute hospital admissions for polio throughout the country, and said the frequency of paralysis was 59% in the unvaccinated, 47% in those who had received one dose prior to the onset of disease, 32% in those who had received two doses, and 23% in those who had received three doses. Even if one were unwary enough to accept these data as reliable, it would follow at most that the effectiveness in preventing paralytic polio was 20% for one dose, 46% for two doses, and 61% for three doses. But the pamphlet states that "Paralytic attack rates have been 75 to 80 per cent lower among persons with one or two inoculations than among the unvaccinated. Preliminary studies among persons with three properly-spaced doses of vaccine suggest that the full course of inoculations may be better than 90 per cent effective against paralytic polio."(21)High spokesmen of the AMA, including its president, Dr. Dwight H. Murray and its chairman of the Board of Trustees, Dr. Gunnar Gunderson joined in this contention. They were supported by Dr. Edgar Martmer, president of the American Academy of Pediatrics and Dr. Malcolm Phelps, president-elect of the American Academy of General Practice. Their articles in the afore-mentioned pamphlet were accompanied by Dr. Salk's own contribution in which he contended that "It has been demonstrated that one dose of vaccine given in 1955 was effective in 60 per cent or more (average 75-80 per cent) (22) This was a reference to the effectiveness of the vaccine in producing response in the way of circulating antibodies, which Dr.. Salk knew not to be measure of effectiveness in preventing the disease.
It is relevant to examine the evidence adduced in favor of the effectiveness of the vaccine. The annual Incidence of poliomyelitis per 100,000 population in the United States from 1910 to 1958 may be indicated as follows:
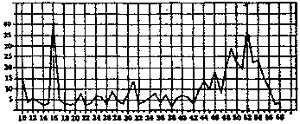
There are two great peaks, one in 1916 and the other in 1952. The rise and fall around the later peak were much less precipitous than in the case of the earlier one. It is, of course, true that polio has diminished considerably since the introduction of the vaccine in 1955; But it diminished even more from 1952 to 1955 before the vaccine; and it decreased far more in the single year from 1916 to 1917, without any vaccine than it did in the first two years with the vaccine. It should, therefore, be obvious that the decrease in itself is no evidence at all-for the efficacy of the vaccine. It is alleged, however, that the decrease has been proportionately greater among the vaccinated than among the unvaccinated, Let us examine the evidence for this.
It is one of the cardinal principles of statistics that no reliable conclusions can be drawn from any samples which are clearly not random. And it is elementary that no sample consisting of volunteers for some procedure, such as vaccination, is random, inasmuch as the very fact that people volunteer indicates a difference from those who do not volunteer, and this difference may affect the outcome of the experiment. As Hill aptly says: "Mothers who bring their babies to be inoculated against an infectious disease may be the more intelligent who take more and better care of their children. Also, such mothers may be more frequently the mothers of single children or of a small family ... A comparison of the inoculated volunteers with the uninoculated non-volunteers, therefore, involves important differences between the groups apart from the state of inoculation, differences which may well influence the relative incidence rates of disease and thereby produce quite misleading comparisons. Without very good evidence of equality such comparisons of volunteers and non-volunteers should not be made."(23)
Secondly, no attempt was made to eliminate personal bias in making the diagnosis of poliomyelitis. There are more than a dozen illnesses due to viruses other than those of poliomyelitis which may be indistinguishable from paralytic polio except by special virus studies. A physician seeing a patient with such paralytic illness at once inquires whether or not the patient has been vaccinated with Salk vaccine, and his diagnosis is very likely to be influenced by the reply. Inasmuch as most physicians have been convinced that triple- vaccination is highly effective, they will make a diagnosis of poliomyelitis if there is no history of vaccination and will make the diagnosis of one of the other diseases if there is a history triple-vaccination.
The entire study from its inception contained sufficient bias to cast grave doubt on its conclusions. And what is even more disturbing is that the aforementioned report, which had been "prepared by members of the Poliomyelitis Surveillance Unit of the United States Public Health Service" and which had originally appeared in mimeographed form for release January 26, 1957, had contained the following paragraph: "Assumptions underlying these analyses introduce several sources of potential bias. Case reports were received through morbidity reporting systems of widely varying accuracy. Overall population figures and vaccine usage figures (particularly for commercial supplies of vaccine) were necessarily estimates. Variations in geographic and age-specific vaccination and attack rates could not be completely accounted for. Risk of exposure was assumed to be equal in the two populations and constant throughout the study period. Although attempts have been made to minimize the effect of these sources of errors, interpretations of the results must necessarily be guarded.(24) This paragraph was omitted in the version given by the National Foundation for Infantile Paralysis. Dr. Ratner said: "It comes as a shock, therefore, to receive in the mail today an informational booklet for physicians which flagrantly violated editorial standards by deleting from a purportedly and otherwise completely reproduced article a short paragraph which discusses the assumptions that are introduced by the authors to guard against the unwary acceptance of the conclusions. The reader is not informed of the deletion. . . The reader can decide for himself whether this deletion is or is not intended to deceive the physician. The fact remains that NFIP has seen fit to decide what physicians of this country shall or shall not read about the Salk vaccine . . . public money which the Foundation holds in trust is expended in a campaign of systematically misleading physicians who have the real responsibility for the public's health."(25)
The conclusion can hardly be avoided that Dr. Rivers and the National Foundation for Infantile Paralysis were responsible for an omission which was indubitably misleading. Medical societies co-operated in the deception.
Surely by now enough time had elapsed since the 1956 epidemic to find out whether any person who did not develop paralytic polio had been triply-vaccinated before the epidemic. Dr. Van Riper's assurance to me 10 months earlier that the answer to my question would be made public was not borne out, and more than three years thereafter it has still not been answered publicly!
An occasional note of disturbance crept into this saga of sunshine and love. Patricia Ann Jennings, an eight-year-old Houston, Texas girl who had been triply-vaccinated with Salk vaccine, died in St. Joseph's Hospital in Houston, and autopsy confirmed that death had been due to paralytic polio.(26) An eight-year-old Chicago boy who had received three injections of "Salk vaccine in 1956 came down with paralytic polio in May of 1957. This news was suppressed more than three months, and was not reported publicly by the Chicago Board of Health until August 26, at which time it was stated that "It was the first case here of paralytic polio contract by a child who" had been given three injections."(27) But for several months after the death, though before it had leaked out, Dr. Bundesen was still promoting the belief that three injections of Salk vaccine never failed to protect.
In spite of all the warning signs the campaign was conducted with full vigor. In November of 1957, AMA mailed two cards and a letter, dated November 25, from its president, David B. Allman, to all physicians in the United States urging. continued vaccination. An evil-minded person who did not know that physicians have no interest in money might be inclined to draw invidious conclusions. . .
Executive Director Raymond Barrows of the National Foundation of Infantile Paralysis was quoted as saying, "A total of about 75,000,000 persons have been given maximum protection with the three recommended Salk shots" but then he added: "A vaccinated child could carry the polio virus home and infect his mother. She might become paralyzed, if she had not been vaccinated."(28)Here was an inadvertent admission that polio vaccine might actually be responsible for, spreading paralytic polio, as suggested almost nine months before by Dr. H. Ratner in a letter to Dr. Hollister, Chief of the Poliomyelitis Vaccination Program, California Department of Public Health. This by no means-proves that vaccination helped spread the disease, however, for a plausible explanation is that vaccination was pushed most in the most heavily infected areas.
About September 1, 1958, there was a case of paralytic polio—and bulbar at that—in a two-and-a-half year-old Chicago boy who had been triply vaccinated; the Chicago Daily News quoted Dr. Bundesen as saying it was the first case in Cook county involving a person who had received all three Salk shots, "and as far as we know it is the first in the united States and possibly in the world."(29) This was six weeks after Dr. Thomas M. Rivers, medical director of the National Foundation for Infantile Paralysis, had admitted that over 200 paralytic cases in trip1y-vaccinated persons had been been reported in the United States alone in 1957;(30) and it was 19 months since the National Foundation for Infantile Paralysis had said there had already been 19 confirmed cases of paralytic polio in the United States in triply vaccinated persons. Perhaps Dr. Bundesen did not know this. But after it was pointed out him, there was no comment from Dr. Bundesen.
But things were not to go entirely smoothly: paralytic polio began to increase in the United States! By September 12 The U.S. Public Health Department reported that there had been 126 cases of paralytic polio reported in triply-vaccinated persons, with six deaths.(31) But this was in its official bulletins, and was not announced to the public at large.
The number of cases in the triply-vaccinated up to September 20 in the years 1957, 1958 was as follows:
1957 1958
paralytic 58 162
non-paralytic 283 325
change from 1957 to 1958
paralytic 179% increase
non-paralytic 14.8% increase
Thus, the number of cases of paralytic polio in the triply-vaccinated had increased by 179%, more than 12 times the increase in non-paralytic polio in the same group. This also strongly suggested the possibility that vaccination was at least partly responsible for the increase in paralytic polio. But these facts were not reported by the press, by the medical journals, or by Dr. Bundesen.
For the six weeks ending September 27 the totals were:
1957 1958
paralytic 522 1014
non-paralytic 838 811
(change from 1957 to 1958
paralytic 94.3% increase
non-paralytic 3.2% decrease(32)
In other words, the incidence of non-paralytic polio had diminished below the previous year's level, but that of paralytic polio kept increasing and was almost double the previous year's, and that was true in almost all parts of the United States.
In the face of this notable failure, U.S. Public Health Service announced that "the Salk vaccine continued to show a high rate of effectiveness—about 87 per cent in 1958, and that it continues to be effective among those who were vaccinated more than three years ago."(33) The House of Delegates of the American Medical Association passed a resolution December 4, 1958, recommending that every physician see to it that every person he examines be vaccinated fully.
The zenith of effrontery was attained just before Christmas when the National Foundation for Infantile Paralysis announced that inasmuch as poliomyelitis had been conquered, they were changing their name to "National Foundation" and turning to what they termed a broadened attack--arthritis and 'birth defects.'"(34)
We have already seen that in November of 1956, Dr. Langmuir had openly admitted that the use of the vaccine in Chicago in 1956 was a great disappointment and that competent health authorities in various countries were agreed that an epidemic could not be stopped in this way.
In January of 1959, the Polio Vaccination Committee of the Chicago Medical Society urged continued vaccination: "There is strong evidence of the continued effectiveness of the vaccine in preventing paralysis—the protection afforded to the age group under 20 years by three shots is calculated as 87%, by two inoculations 74%, and by one inoculation 35%. ... A year ago this committee recommended that a fourth 'booster' dose be given to all persons under 21 who had completed the third inoculation more than one year previously. It is hoped that this 'booster" dose will raise the prevention afforded from the present calculated 87% to 100%."(35) This defies polite comment, for the Committee knew perfectly well that in the two years 1957-1958 there had been at least 58 cases of paralytic polio among relatively few persons who had been quadruply-vaccinated, so it was absolutely certain that four doses were very far from 100% effective. In February, all Illinois physicians were urged by the Illinois State Medical Society and the American Academy of Pediatrics to give Salk vaccine to all patients between the ages of two months and 40 years, with the incorrect statement that "all authorities agree on the advisability of the 4th injection." As recently as the preceding October the U.S. Public Health Service had said there was no reason to change from the three-dose schedule,(36) and as recently as December that it was protective for more than three years.(37) The Illinois State Medical Society, while admitting that "Polio is not licked," said that the Salk vaccine had "the best record of any immunizing agent in general use;" this was an incorrect statement, for it was common knowledge that several other generally used immunizing agents, such as those against smallpox and diphtheria, had decidedly better records.
In March, 1959, Mr. Basil O'Connor, president of the National Foundation, in urging against a united fund for all charities, said March of Dimes had raised more than 500 million dollars in 15 years. He said further: "The result was the Salk vaccine. Because of it, no child in this country or anywhere else in the world need ever again suffer one disease—polio."(38) On March 13 it was reported that since January 1 there had been 193 cases of polio—of which 135, or 70%, were paralytic—compared with a total for the same period 1958 of 153 cases of which 87 or 57% were paralytic. The paralytic cases were 55% higher 1959 than in 1958; At this very time the Chicago Medical' Society said: "A 30-year battle against diphtheria, and a 100-year war against smallpox cannot claim results comparable to those obtained in a two year fight against polio."(39) This was true only of the financial results.
On April 26, 1959, Mr. O'Connor admitted that(40)in the first quarter of this year there were 83% more cases" of paralytic polio, than in the first quarter of 1958; three weeks later(41) this increase had grown to more than 100%. A month later Dr. Bundesen said: "Every parent has the responsibility to see that his children get the shots. We now have a weapon to fight polio. That weapon is the Salk vaccine. The best known method to prevent a polio outbreak is to keep the community thoroughly inoculated. If that is done, there will be no outbreak. . . Widespread use of the vaccine is undoubtedly the reason why not a single case of polio has been reported here since January 1." (42)
In a round-table discussion at the Minnesota State Medical Association it was revealed(43) that although 90% of children under six years of age in Israel had been given Salk shots there was at that time an epidemic of polio in Israel.
On June 19 the U.S. Public Health Service announced that for the first 23 weeks of 1959 there were 452 cases of paralytic polio as compared to 231 in the first 23 weeks in 1958; this was an increase of 95.7%. A week later the Public Health Service refused to license the Albert Sabin live virus vaccine, to be given orally, on grounds it was "still in the experimental stage," and urged the public to get the "protection" of the Salk vaccine.(44) It was casually stated that the Salk vaccine is made from a mixture of killed polio viruses and is given in a series of four injections, with no reference to the fact that for the past four years assurance had been given the public that three injections were highly protective.
In the face of a marked increase in paralytic poliomyelitis for the past two years in the United States, Dr. A. D. Langmuir of the U.S. Department of Health, Education and Welfare still wrote concerning the Salk vaccine as follows: "Completion of the immunization program should lead to the essential elimination of paralytic poliomyelitis from the United States. . . The marked downward trend of poliomyelitis in the past four years is due in large part to the immunization program . . . the complete elimination of the paralytic form of the disease ... is within reach."(45)The Journal of the American Medical Associationz published this without comment.
The considerable increase in paralytic poliomyelitis in the United States during the past two years, despite the progressivedecrease in the number of the most susceptible persons (those under 40) who have not yet been triply-vaccinated, does not prove that the vaccine is valueless. But the evidence, biased as it is in favor of the vaccine, suggests that it may be of little or no value. Even more, it suggests the distinct possibility that the vaccine may actually be at least partly responsible for the increase by producing carriers who spread the disease.
[No Footnote #1.]
2. Meier, P., Safety Testing of Poliomyelitis Vaccine. Science, 125:067, 1957.
3. Ibid.
4. Brownlee, K. A., Statistics of the 1954 Polio Vaccine Trials, Journ. Amer. Statist. Assoc, 50: 1005, 1955.
5. Journal of the American Medical Association, 155:205, 1954.
6. Brownlee, K. A. Statistics of the 1954 Polio Vaccine Trials, Journ. Amer. Statist. Assoc, 50; 1005, 1955.
7. Ratner, H., Poliomyelitis Vaccine, Journ. Amer. Med. Assoc, 160:31, 1956.
8. Chicago Daily News, June 1956.
9. Ibid.
10. Chicago Sun-Times, July 27, 1956.
11. Ibid., August 9, 1956.
12. Ibid., September 28, 1956.
13. Ibid., September 30, 1956.
14. Ibid., November 29, 1956.
15. New York Times, January 4. 1957.
16. Chicago Daily News, March 14, 1957.
17. Ibid., March 14, 1957.
18. World Health Organization, Technical Report Series, No. 145. Expert Committee on Poliomyelitis. Second Report, p. 34, Geneva. 1958.
19. Information for Physicians on the Salk Poliomyelitis Vaccine, No. 4, February 1957.
20. Ibid., p. 27.
21. National Foundation for Infantile Paralysis, Information for Physicians on the Salk Poliomyelitis Vaccine, No. 4, p, 2, February 1957.
22. Ibid., p. 18.
23. Hill, A. B., Principles of Medical Statistics, p. 18. Oxford University Press, New York I955.
24. Ratner, H., Letter to the editors of sundry medical journals, February 28, 1957.
25. Ibid.
26. Chicago Sun-Times. August 25, 1957.
27. Ibid., August 27, 1957.
28. Chicago Sun-Times, November 20, 1957.
29. Chicago Daily News, September 2, 1958.
30. Rivers, T. M., Letter to the editor, Journ. Amer. Med. Assoc, 167:1551, 1958.
31. Poliomyelitis Surveillance Report No. 151, September 12, 1958.
32. Ibid., No. 154, October 3, 1958.
33. Chicago Sun-Times, December 12, 195S.
34. Journal of the American Medical Association, 259:1275, 1958 (December 25).
35. Chicago Medical Society Bulletin, p. 519, January 17, 1959.
36. U.S. Department of Health, Education and Welfare, Communicable Disease Center Poliomyelitis Surveillance Report, Supplement No. 16, 1958.
37. Chicago Sun-Times, December 12, 1958.
38. Ibid., March 1959.
39. Chicago Medical Society Bulletin, p. 704; March 21, 1959.
40. Chicago Tribune. April 27, 1959.
41. Chicago Daily News, May 16, 1959.
42. Chicago Tribune, May 24, 1959.
43. Chicago Daily News, March 29, 1959.
44. Chicago Sun-Times, June 26. 1959.
45. Langmuir, A. D., Progress in Conquest of Paralytic Poliomyelitis. Journ. Amer. Med. Assoc, 171:271, 1959.

